The Foundation of Food Safety
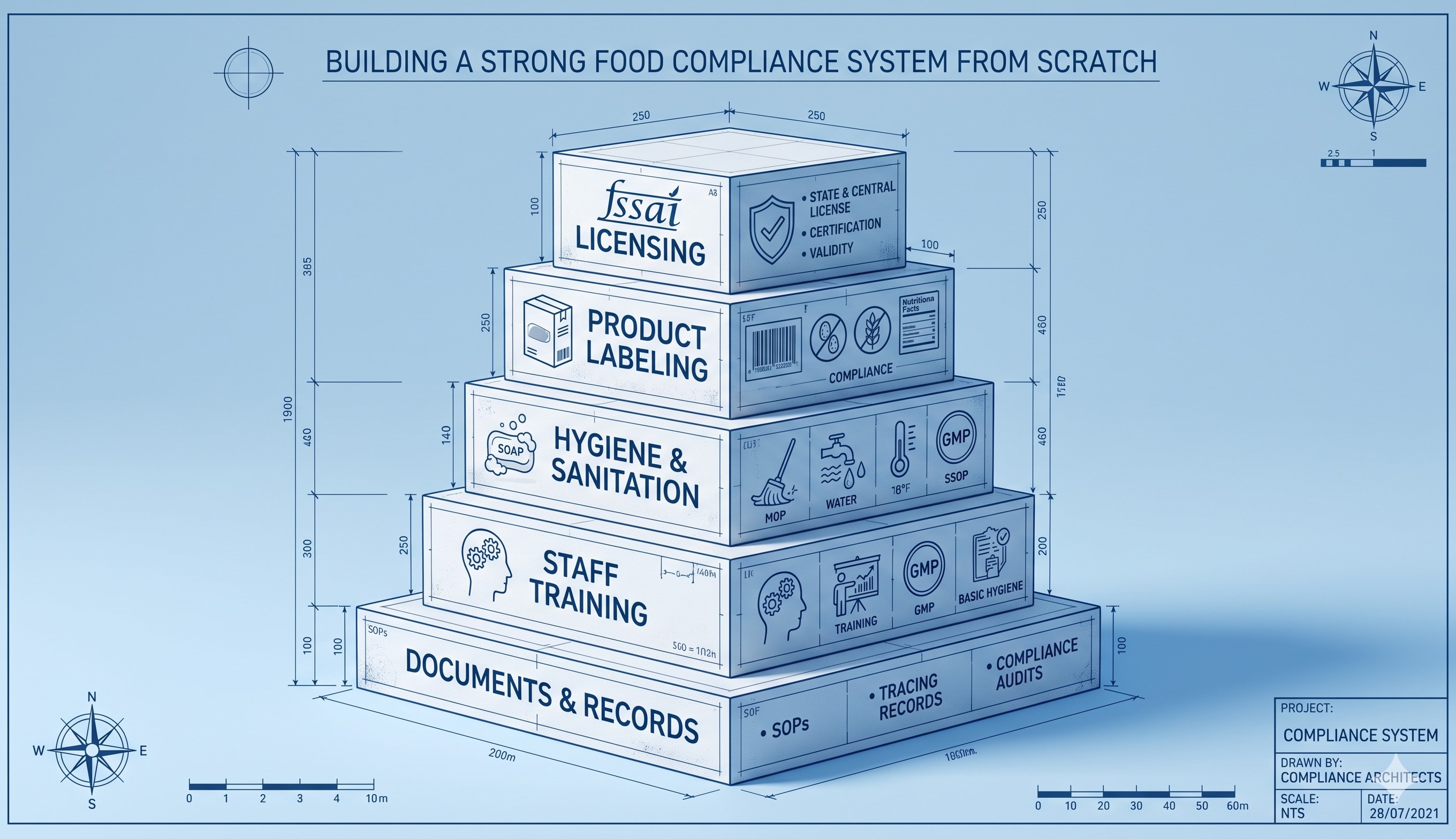
Building a strong food compliance system from scratch is the most critical investment an emerging nutraceutical or food business can make in the Indian market. A robust compliance framework is not simply about passing an initial FSSAI inspection; it is about embedding a culture of safety, quality, and regulatory adherence into the very DNA of your daily operations. Whether you are setting up a manufacturing plant or a distribution network, a well-architected system protects consumers from health hazards while shielding your brand from catastrophic legal liabilities. From raw material procurement to final product dispatch, every step of the supply chain must be strictly governed by standardized, verifiable protocols. FSSAI mandates that Food Business Operators (FBOs) actively demonstrate their commitment to safety through documented systems that preemptively identify and neutralize potential hazards. By prioritizing compliance from day one, businesses build a sustainable operational model that can easily adapt to scaling production volumes and evolving regulatory demands in India's highly competitive food sector.
Implementing FSMS and HACCP Principles
The core of any resilient compliance architecture is a fully integrated Food Safety Management System (FSMS), heavily anchored in Hazard Analysis and Critical Control Point (HACCP) principles. Developing an FSMS requires a meticulous, scientific evaluation of your entire manufacturing process to pinpoint exact locations where biological, chemical, or physical contamination could occur. Once these critical control points are identified, rigorous preventive measures and continuous monitoring protocols must be established and documented. This involves creating highly detailed Standard Operating Procedures (SOPs) that dictate exactly how machinery is sanitized, how raw ingredients are stored, and how finished products are quarantined and tested before release. FSSAI requires this documentation to be thorough, up-to-date, and readily available for unannounced audits. Implementing a strong FSMS transforms compliance from a reactive scramble into a proactive, predictable operational rhythm. It ensures that every batch of nutraceuticals or food products manufactured consistently meets the highest safety standards without fail, establishing unbreakable consumer trust.
Documentation and Traceability Standards
A compliance system is only as strong as the documentation that supports it. FSSAI regulations demand absolute transparency and traceability throughout the entire supply chain. Building a system from scratch requires implementing robust record-keeping practices that can instantly trace any finished product back to its specific batch of raw ingredients, the exact date of manufacture, and the personnel involved. This level of traceability is essential for executing rapid, targeted product recalls if a safety issue ever arises. Your documentation architecture must include comprehensive supplier audit reports, daily production logs, equipment maintenance schedules, and third-party laboratory testing certificates for both ingredients and water quality. Transitioning from scattered paperwork to a centralized, digitally managed documentation system ensures that vital information is never lost and is instantly retrievable during regulatory inspections. Pristine record-keeping not only proves your adherence to FSSAI standards but also provides invaluable data for continuous process improvement and operational efficiency analysis.
Employee Training and Hygiene Protocols
The most sophisticated compliance system will instantly collapse if the personnel operating it are not adequately trained. Building compliance from the ground up necessitates mandatory, rigorous, and ongoing training programs for all employees involved in food handling, processing, and management. FSSAI’s Schedule 4 guidelines place immense emphasis on Good Hygiene Practices (GHP), detailing strict requirements for personal cleanliness, protective clothing, and behavioral protocols within the manufacturing facility. Employees must deeply understand the "why" behind the SOPs, recognizing how their individual actions directly impact food safety and regulatory compliance. Regular workshops on cross-contamination prevention, proper sanitization techniques, and hazard identification are essential. Furthermore, FBOs are required to maintain records of routine medical check-ups for all staff to prevent the transmission of communicable diseases. Cultivating an educated, deeply responsible workforce ensures that your compliance protocols are actively enforced on the factory floor every single minute of the operational day.
Internal Auditing and Continuous Improvement
A static compliance system is a vulnerable compliance system. The final pillar of building a strong framework is establishing a rigorous schedule of internal audits designed to relentlessly test the efficacy of your protocols. Internal audits simulate FSSAI inspections, allowing you to proactively identify vulnerabilities, outdated procedures, or lapses in employee adherence before they evolve into regulatory violations. These audits must be conducted objectively, covering every aspect of the facility from waste management and pest control to labeling accuracy and storage temperatures. Any deviations discovered during an audit must trigger immediate Corrective and Preventive Actions (CAPA), which are thoroughly documented and verified for effectiveness. This cycle of continuous monitoring and improvement guarantees that your compliance system remains agile, responsive to new FSSAI notifications, and capable of mitigating unforeseen risks. An aggressive internal audit strategy is the hallmark of a mature, legally secure nutraceutical operation dedicated to long-term market dominance.
The Devastating Cost of System Failures
Attempting to scale a food business on a weak or non-existent compliance foundation is a recipe for operational disaster and profound financial loss. The risks of cutting corners during the system-building phase are immense; regulatory authorities show zero leniency for negligence that endangers public health. A single failure in your unverified supply chain or a lapse in undocumented hygiene protocols can lead to widespread product contamination. When FSSAI inspectors uncover systemic failures, the consequences are swift and severe: immediate suspension of manufacturing operations, massive financial penalties, and mandatory destruction of inventory. Furthermore, the reputational damage resulting from a public safety incident or a forced recall is often irreversible, destroying investor confidence and permanently alienating consumers. The bureaucratic nightmare of attempting to retrofit a compliance system after a regulatory violation is vastly more expensive and disruptive than building it correctly from the start. Independent system architecture without expert guidance is a catastrophic risk.
Partner with Food 1947 for Bulletproof Systems
Designing and executing a flawless compliance system from scratch requires specialized expertise that goes far beyond basic business management. It demands a deep, technical understanding of FSSAI regulations and industry-specific best practices. Partner with Food 1947 to completely eliminate the guesswork, risk, and overwhelming stress of building your regulatory infrastructure. Our elite consultants specialize in architecting bulletproof, end-to-end Food Safety Management Systems tailored specifically to the Indian nutraceutical and food sectors. We handle the heavy lifting: drafting meticulous SOPs, designing robust traceability matrices, conducting comprehensive staff training, and pre-auditing your facilities to guarantee total FSSAI readiness. Do not jeopardize your investment by attempting to decipher complex compliance frameworks alone. We ensure your business launches on a foundation of absolute legal certainty and operational excellence. Secure your brand’s future and accelerate your path to market dominance. Contact Food 1947 today for a strategic consultation and let us build your impenetrable compliance system.





