The Frontline of Consumer Trust
The product label is the absolute frontline of consumer trust and the primary battleground for regulatory compliance in the Indian food and nutraceutical market. Understanding the 7 Must-Have Elements on Every Food Label is not merely a design exercise; it is a strict legal requirement mandated by the Food Safety and Standards Authority of India (FSSAI). A label serves as the direct communication channel between the manufacturer and the consumer, dictating everything from product identity to critical safety warnings. For dietary supplements and specialized foods, this real estate becomes even more complex, requiring precise scientific terminology and strict adherence to formatting guidelines. The FSSAI has developed exhaustive packaging and labeling regulations designed to eradicate misleading claims, prevent allergen exposure, and ensure absolute transparency regarding nutritional content. Every millimeter of your packaging must be meticulously audited to ensure that mandatory elements are displayed prominently, clearly, and in exact accordance with the law, forming the bedrock of your brand's legal market presence.
Product Name and Veg/Non-Veg Declaration
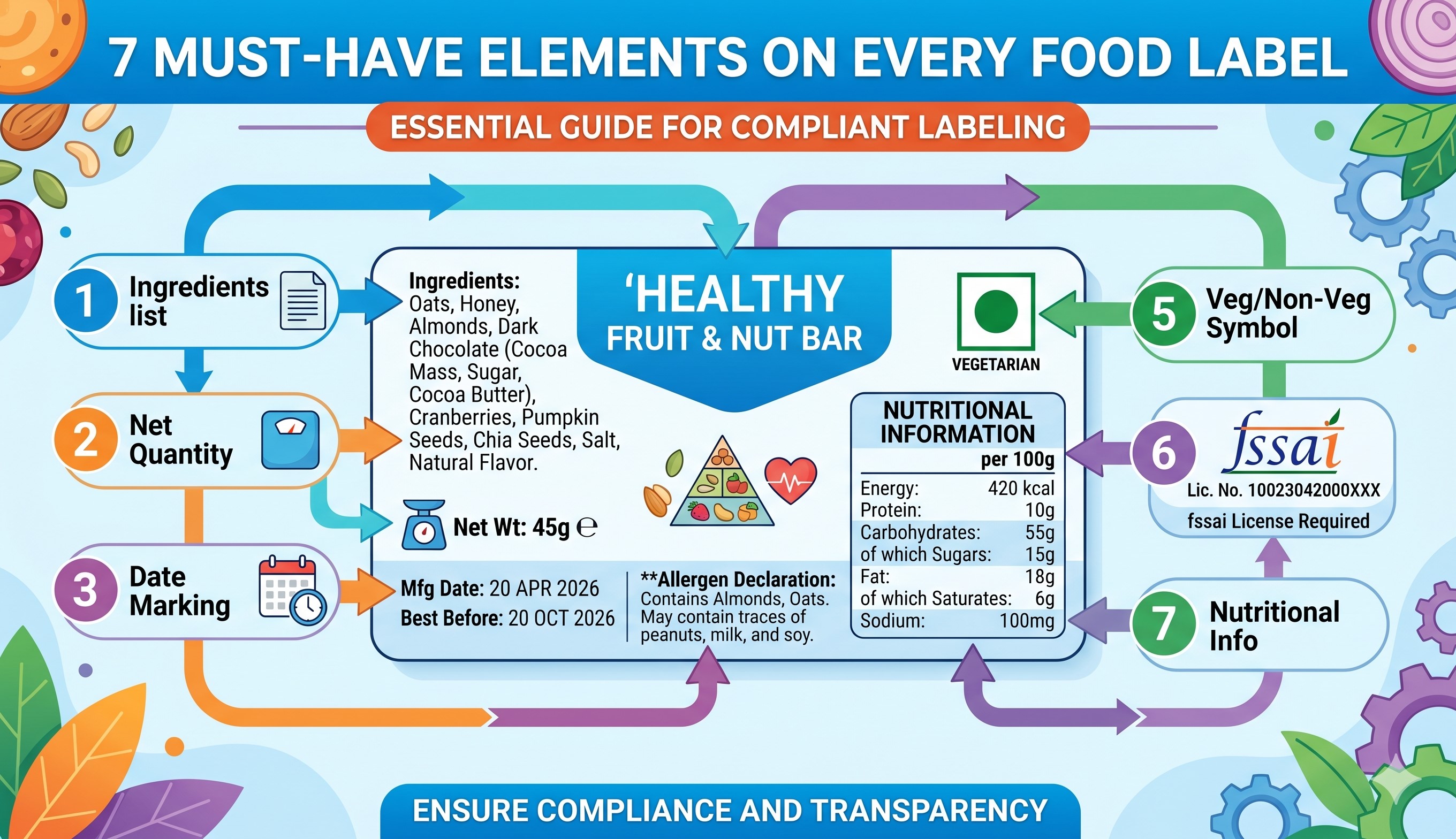
The first mandatory element is the clear, unambiguous name of the food product, which must accurately reflect its true nature and not mislead the consumer. For nutraceuticals, this often means including specific classifications like "Health Supplement" or "Food for Special Dietary Use" directly adjacent to the brand name. Equally critical is the mandatory vegetarian or non-vegetarian declaration. FSSAI requires a highly specific logo—a green circle inside a green square for vegetarian, and a brown triangle inside a brown square for non-vegetarian—placed prominently on the principal display panel. The exact dimensions and placement of this logo are strictly regulated based on the total surface area of the package. A missing, miscolored, or incorrectly sized veg/non-veg logo is one of the most common labeling violations and results in immediate product rejection by retail chains and regulatory inspectors alike, halting your distribution instantly.
Comprehensive List of Ingredients
The second crucial element is the complete list of ingredients, presented in descending order of their weight or volume at the time of manufacture. This section demands absolute precision, particularly for nutraceutical formulations that utilize complex botanical extracts, standardized active compounds, and specialized excipients. FSSAI mandates that specific names of ingredients must be used, rather than generic categories, and any added water must be declared if it exceeds a certain threshold. Furthermore, the exact percentages of key characterizing ingredients highlighted on the front of the pack must be explicitly detailed in the ingredient list. For example, if a product claims to be a "Turmeric Extract Supplement," the exact concentration of curcuminoids must be verified and listed. Attempting to hide proprietary blends by obfuscating individual components is a direct violation of FSSAI transparency rules and triggers severe regulatory scrutiny and penalization.
Nutritional Information and RDA
The third non-negotiable element is the nutritional information panel, which must clearly state the energy value (calories), protein, carbohydrate, total sugar, added sugar, and fat content per 100g, 100ml, or per serving. For the nutraceutical sector, this requirement escalates significantly. Supplements must detail the exact quantities of every vitamin, mineral, and amino acid present, alongside their respective Recommended Dietary Allowance (RDA) percentages specifically calculated for the Indian population. FSSAI has strict guidelines on how these values are calculated, formatted, and presented in tabular form. Any variation between the declared nutritional values and actual laboratory test results can be classified as adulteration or misbranding. Ensuring the absolute mathematical accuracy of your nutritional panel requires rigorous independent lab testing and meticulous formatting to meet FSSAI’s exact visual and scientific standards, preventing costly product recalls and legal disputes.
Additives, Allergens, and Warnings
Elements four, five, and six encompass food additives, allergen declarations, and mandatory safety warnings. All artificial colors, synthetic flavors, and chemical preservatives must be declared using their specific class titles and internationally recognized INS (International Numbering System) numbers. FSSAI is incredibly strict regarding permissible limits for additives in specific food categories. Furthermore, the presence of common allergens—such as peanuts, tree nuts, soy, milk, eggs, or gluten—must be highlighted conspicuously to prevent life-threatening consumer reactions. Finally, the label must feature specific warning statements mandated by law. For nutraceuticals, this includes disclaimers like "Not for Medicinal Use," warnings against exceeding the recommended daily dose, and cautions for pregnant women or children. Omission of a single mandatory allergen alert or regulatory warning transforms your product into a significant public health hazard, exposing the company to massive legal liability and criminal prosecution.
The Severe Penalties for Misbranding
The final must-have elements include the manufacturer's address, FSSAI license number, net quantity, batch number, and essential date marking. Attempting to design and approve food labels without expert regulatory oversight introduces catastrophic risk. FSSAI considers any deviation from their exhaustive labeling laws as 'misbranding.' The penalties for launching a misbranded product are severe, encompassing immediate stop-sale orders, forced market recalls at the manufacturer's expense, and financial fines that can reach up to three lakh rupees per violation. If a labeling error involves an undeclared allergen or a false health claim, the authorities may initiate criminal proceedings and permanently revoke your manufacturing license. The complexity of font size minimums, contrast ratios, and claim substantiation makes DIY label design a dangerous gamble. A single typo or a misunderstood regulation can render thousands of freshly printed packaging units entirely useless, causing massive financial hemorrhaging and critical delays in your product launch timeline.
Partner with Food 1947 for Flawless Labels
Achieving 100% accurate, legally compliant food labeling is a highly technical discipline that demands specialized regulatory expertise. Do not risk your brand’s reputation or face devastating FSSAI penalties by relying on graphic designers who lack deep knowledge of Indian food laws. Partner with Food 1947 to guarantee your labels are utterly flawless and market-ready. Our elite compliance experts conduct microscopic audits of your packaging artwork, meticulously validating every ingredient, RDA calculation, font size, and health claim against the latest FSSAI gazette notifications. We eliminate the immense stress and financial risk of regulatory rejections, ensuring your product launches smoothly and legally. Protect your bottom line, accelerate your market entry, and secure absolute peace of mind by letting seasoned professionals handle your label compliance. Contact Food 1947 today for a comprehensive label review and let us fortify your products against all regulatory risks in the competitive Indian market.





